Beyond Monitoring. A Governance System for Nutraceutical Integrity.
Nutracovigilance® transforms safety from passive reporting into structured leadership, accountability, and system-wide oversight — across the full lifecycle of nutraceutical products.
Architected by
Dr. Ronni Shino, PhD, DBA (cand.) · Shinlaza Inc.
What Is Nutracovigilance®?
"Nutracovigilance® represents a managerial and governance framework that extends beyond adverse event monitoring to encompass the strategic oversight, integration, and accountability of safety, quality, and regulatory intelligence across the full lifecycle of nutraceutical products."
This is not a clinical concept. It is not a variation of pharmacovigilance. Nutracovigilance is a governance doctrine — a structured architecture of leadership, accountability, and cross-functional integration designed to address the systemic failures that no monitoring system alone can resolve.
Nutracovigilance IS
Nutracovigilance is NOT
A Managerial Failure, Not a Data Problem
The nutraceutical industry does not lack data. It lacks the governance structures, leadership integration, and accountability systems required to act on that data with strategic coherence. This is a managerial failure — not a clinical limitation.
"The problem is not the absence of data — it is the absence of governance."
Governance Structures
The industry operates without formal governance architectures that assign accountability across the product lifecycle. Safety intelligence exists in silos — disconnected from strategic decision-making.
Leadership Integration
Safety and quality functions are treated as operational departments rather than strategic leadership domains. Executive accountability for nutraceutical integrity remains structurally undefined.
Accountability Systems
There are no standardized mechanisms for cross-functional accountability. Manufacturers, regulators, and market actors operate without shared governance frameworks or enforceable oversight protocols.
Nutrivigilance vs. Nutracovigilance
The distinction is not semantic. It is structural. Nutrivigilance operates as a detection instrument. Nutracovigilance operates as a governance architecture — encompassing the full strategic, operational, and executive dimensions of nutraceutical integrity.
| Dimension | Nutrivigilance | Nutracovigilance |
|---|---|---|
| Approach | Reactive | Proactive + Preventive |
| Scope | Post-market surveillance | Full product lifecycle |
| Function | Monitoring | Governance |
| Role | Compliance | Leadership |
| Structure | Fragmented | Integrated |
| Accountability | Undefined | Structured & Assigned |
| Decision Layer | Operational | Executive & Strategic |
Five Pillars of Governance Architecture
The Nutracovigilance framework is structured around five integrated governance pillars. Each pillar addresses a distinct dimension of organizational accountability — together forming a comprehensive architecture for nutraceutical integrity.
Surveillance Intelligence
Overview
A systematic architecture for collecting, processing, and interpreting safety signals across all product lifecycle stages — from formulation through post-market deployment.
Real-World Relevance
Manufacturers operating in multiple regulatory jurisdictions require unified signal detection systems that aggregate adverse event data, consumer feedback, and scientific literature into actionable intelligence.
Strategic Implication
Organizations that treat surveillance as a governance input — rather than a reporting obligation — gain early warning capability and strategic positioning advantage in regulatory environments.
Manufacturing & Quality Governance
Overview
A structured oversight model that embeds quality assurance and safety accountability into manufacturing leadership, not merely into quality control departments.
Real-World Relevance
Supply chain complexity, contract manufacturing relationships, and ingredient sourcing variability demand governance frameworks that assign executive accountability for quality outcomes.
Strategic Implication
Quality governance at the leadership level transforms manufacturing from a cost center into a strategic differentiator, enabling premium positioning and regulatory resilience.
Regulatory Interface Integration
Overview
A proactive model of regulatory engagement that positions organizations as governance partners rather than compliance subjects — anticipating regulatory evolution and shaping it.
Real-World Relevance
As nutraceutical regulation converges globally, organizations without dedicated regulatory intelligence functions face strategic exposure to market access disruption and enforcement action.
Strategic Implication
Regulatory interface integration converts compliance costs into strategic intelligence assets, enabling organizations to lead regulatory conversations rather than respond to them.
Market & Consumer Risk Monitoring
Overview
A continuous market intelligence function that monitors consumer behavior, competitive dynamics, and emerging risk signals — integrating market data into governance decision-making.
Real-World Relevance
Social media, e-commerce platforms, and direct-to-consumer channels generate real-time risk signals that traditional post-market surveillance systems are structurally unable to capture.
Strategic Implication
Organizations that govern market risk proactively can protect brand equity, anticipate consumer safety concerns, and position themselves as responsible market leaders.
Executive Oversight & Accountability
Overview
A formal governance structure that assigns executive-level accountability for nutraceutical integrity — embedding safety and quality leadership into the organizational hierarchy.
Real-World Relevance
Boards, CEOs, and COOs in the nutraceutical sector currently lack defined governance responsibilities for product integrity. This structural gap creates organizational liability and strategic risk.
Strategic Implication
Executive accountability frameworks transform nutraceutical governance from a departmental function into a board-level strategic imperative — the foundation of institutional credibility.
From Detection to System Control
"Nutrivigilance is a radar system.
Nutracovigilance is air traffic control."
Radar System
Detects signals. Reports positions. Identifies anomalies. Passive by design — it observes but does not act.
Air Traffic Control
Manages the system. Coordinates actors. Assigns accountability. Active by design — it governs outcomes, not just observations.
The governance model of Nutracovigilance does not replace surveillance — it contextualizes it. Signal detection is a necessary input. But governance is the system that decides what to do with those signals, who is accountable for acting on them, and how that action is structured, documented, and evaluated.
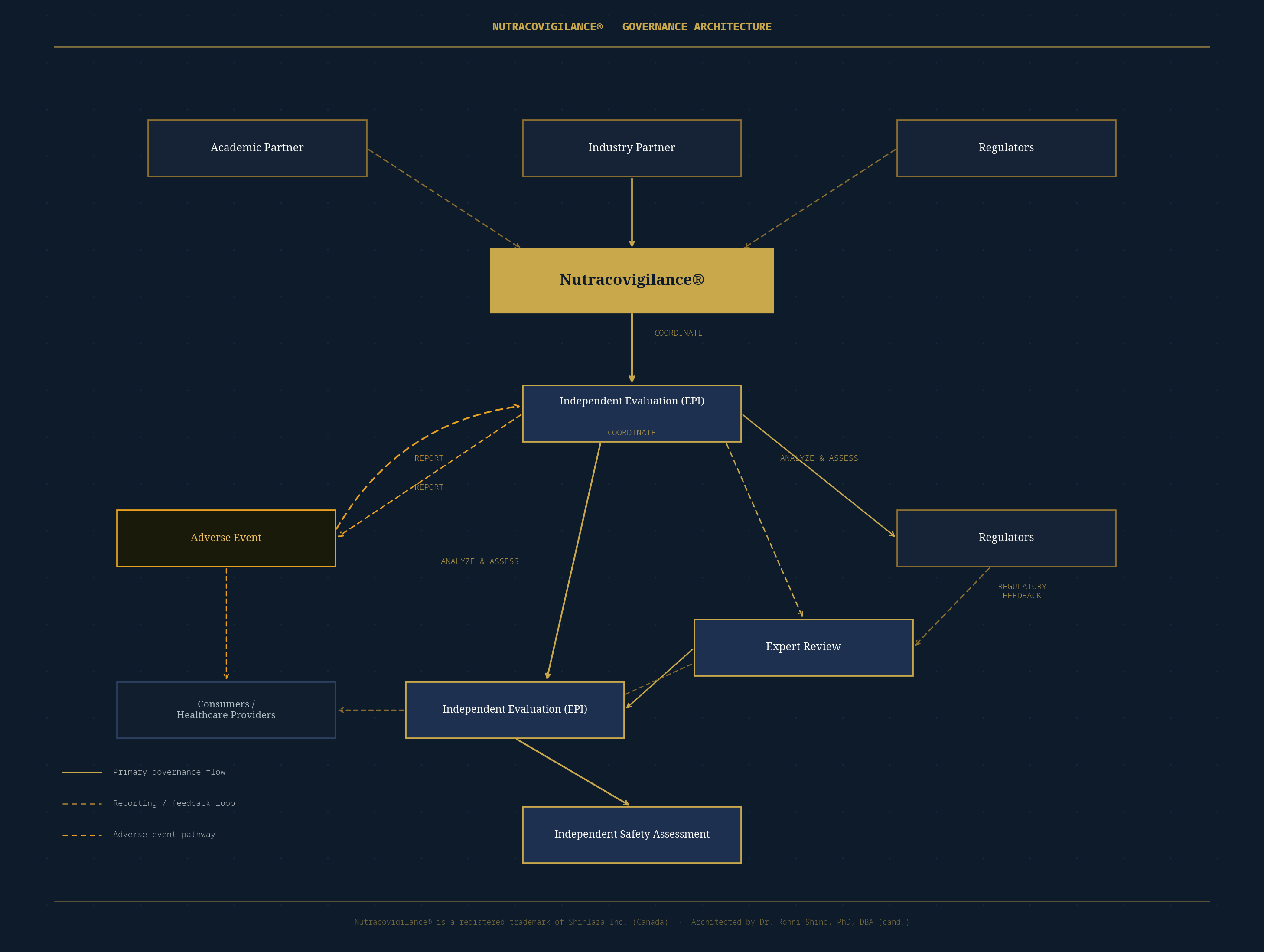
The diagram below illustrates the full governance architecture of Nutracovigilance® — showing how the framework coordinates academic partners, industry actors, regulators, independent evaluation bodies, and safety assessment processes into a unified, accountable system.
Nutracovigilance® Governance Architecture · Shinlaza Inc. · Dr. Ronni Shino, PhD, DBA (cand.)
A Governance Doctrine for Executive Leadership
Nutracovigilance® addresses four dimensions of organizational failure that no clinical intervention, regulatory update, or monitoring technology can resolve. These are structural problems that require structural solutions. Developed through concurrent doctoral research at SSBR (Switzerland) and ESGCI (France), this framework represents the convergence of management theory and nutraceutical governance practice.
Strategic Leadership Problem
The failure to govern nutraceutical safety is, at its core, a leadership failure. It reflects an absence of executive ownership, strategic prioritization, and organizational accountability at the highest levels of the enterprise.
Governance Gap
The nutraceutical sector operates without a standardized governance architecture. There are no universally adopted frameworks that assign accountability, define oversight responsibilities, or integrate safety intelligence into strategic decision-making.
Systems Integration Failure
Safety, manufacturing, regulatory, and market functions operate as disconnected silos. The absence of cross-functional integration means that critical intelligence fails to reach the decision-makers who need it — and accountability fails to reach those who bear it.
Not a Clinical Limitation
This is not a problem that more clinical research, better biomarkers, or improved analytical methods will solve. The data exists. The science advances. What is absent is the governance architecture to act on it with coherence, accountability, and strategic intent.
"This is not a concept. This is not a theory. This is a new governance standard."
Dr. Ronni Shino
Shinlaza Inc.
Built for Leaders Who Govern
Nutracovigilance is designed for those who bear responsibility for nutraceutical integrity at the organizational, regulatory, and policy level. It is a framework for decision-makers, not observers.
Dr. Ronni Shino
PhD, DBA (cand.)
Managing Director, Shinlaza Inc.
Dr. Ronni Shino is the originating architect of the Nutracovigilance® governance framework — a doctoral-level construct that redefines how the nutraceutical industry approaches safety, accountability, and strategic oversight. The framework is the subject of active doctoral research at two leading international institutions simultaneously, reflecting its depth of theoretical grounding and real-world applicability.
As Chief Architect of the Mechanism Drive Biomedical Intelligence Framework and Managing Director of Shinlaza Inc., Dr. Shino operates at the intersection of biomedical science, governance theory, and executive leadership. The Nutracovigilance® framework represents the convergence of these domains into a structured, institutionalizable governance doctrine.
The Canadian trademark for Nutracovigilance® is held by Shinlaza Inc., affirming the framework's status as an original intellectual property contribution to the governance of nutraceutical products globally. Dr. Shino is currently engaged in the active institutionalization of the framework across multiple jurisdictions, with formal country engagements underway and a structured global adoption roadmap in execution.
Active Doctoral Research
"The Future of Nutracovigilance: Business Strategy, Healthcare Integration, and the Changing Landscape of Dietary Supplements"
Pursued concurrently at SSBR (Switzerland) — PhD & ESGCI (France) — DBA
Academic
Professional
Research
Organizational Affiliations
Nutracovigilance® is a registered trademark of Shinlaza Inc. (Canada). All rights reserved.
Institutionalizing a New Global Standard
Nutracovigilance® is not a concept awaiting adoption — it is a governance framework in active global deployment. Under the leadership of Dr. Ronni Shino and Shinlaza Inc., formal country engagements are currently underway across eight jurisdictions, with a structured roadmap targeting 15 countries by 2030 and full global institutionalization thereafter.
Active Jurisdictions — 2025
Global Adoption Roadmap
Trademark secured. Framework architecture finalized. Active engagement initiated across 8 jurisdictions. Target: onboard 3 countries under formal governance adoption.
Formal adoption by regulatory bodies, industry associations, and executive institutions across initial country cohort. Doctoral research publications released.
15 countries operating under the Nutracovigilance® governance framework. Framework recognized as the international standard for nutraceutical governance architecture.
Full global expansion. Nutracovigilance® embedded in executive education curricula, regulatory policy frameworks, and multinational corporate governance structures worldwide.
"The institutionalization of Nutracovigilance® is not a future ambition. It is an active process — country by country, institution by institution, governance structure by governance structure."
— Dr. Ronni Shino
The Future of Nutraceutical Governance
The trajectory of the nutraceutical industry is toward greater regulatory complexity, higher consumer expectations, and increased organizational accountability. Nutracovigilance is the governance architecture that will define how responsible organizations navigate that trajectory.
Institutional Standardization
Nutracovigilance will become the reference framework for governance standardization across the global nutraceutical sector — adopted by regulatory bodies, industry associations, and multinational manufacturers as the baseline for organizational accountability.
Executive Curriculum Integration
Business schools, doctoral programs, and executive education institutions will integrate Nutracovigilance as a case study in governance architecture — bridging the gap between management theory and nutraceutical industry practice.
Regulatory Convergence
As global regulatory frameworks for nutraceuticals converge, Nutracovigilance provides the governance infrastructure that organizations will require to navigate multi-jurisdictional compliance with strategic coherence and executive accountability.
Digital Governance Integration
The integration of AI-driven surveillance intelligence, real-time market monitoring, and automated regulatory interface systems will require a governance architecture capable of managing digital complexity — a domain where Nutracovigilance provides the foundational framework.
The Nutracovigilance Governance Framework: A White Paper
A comprehensive academic and practitioner document presenting the theoretical foundations, structural architecture, and implementation pathways of the Nutracovigilance governance framework. Designed for executives, regulatory leaders, and doctoral researchers.
Govern with Intention
Nutracovigilance is not a passive framework. It is an active governance doctrine designed for organizations and leaders who are ready to move beyond compliance and into genuine accountability.
Adopt the Framework
Implement the Nutracovigilance governance architecture within your organization. We provide structured onboarding, executive workshops, and implementation support for leadership teams.
Partner in Governance
Collaborate with us to advance the Nutracovigilance framework through research partnerships, institutional adoption, and cross-sector governance initiatives.
Request Advisory
Engage directly with the framework's architects for executive advisory, doctoral consultation, regulatory strategy, or organizational governance assessment.
For institutional inquiries, doctoral collaboration, or executive advisory engagement, contact the Nutracovigilance governance team directly.